The campaign “The Elements of Excellence” represents everything that defines the company: from the vision, the people, the history, the customers, the quality to every component and, of course, every product.
Like a kaleidoscope that constantly creates new, impressive images through the combination of colours and shapes, KaVo continuously develops new technologies and products that redefine and improve dentistry.
KaVo Product Innovations 2025
KaVo Highlights & News
KaVo Highlights & News
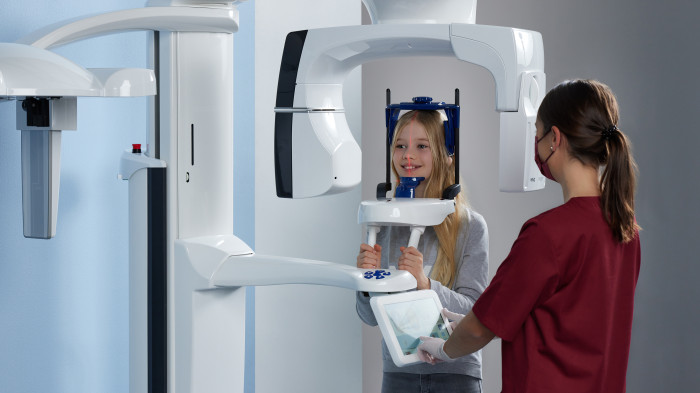
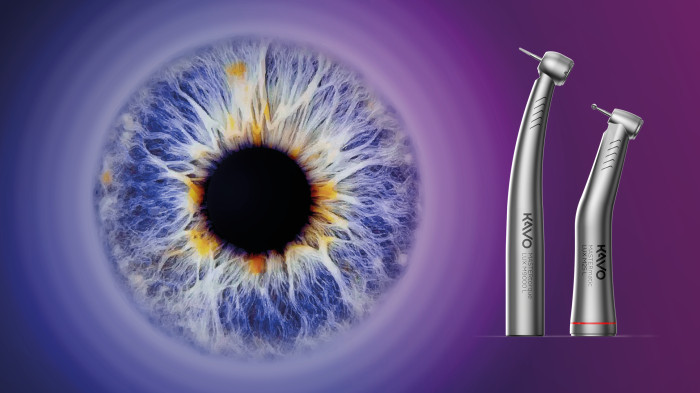
SEE IT! Get the best possible view with KaVo instruments.
Take the opportunity NOW to experience and secure first-class quality, precision and above all: the best view with KaVo!
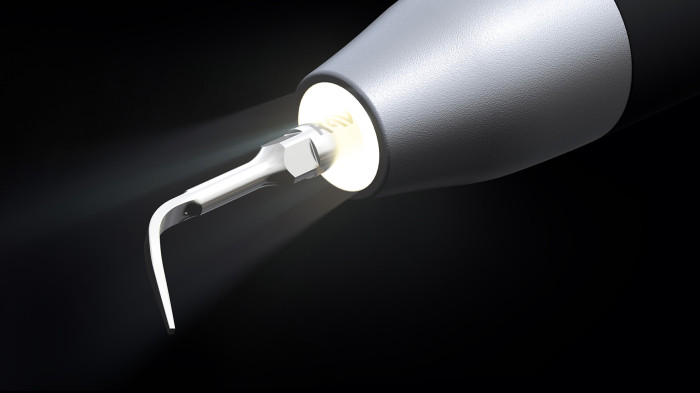
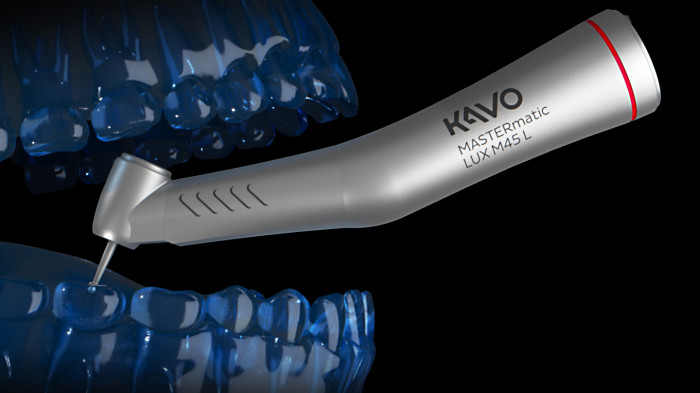
MASTERmatic LUX M45 L high-speed contra-angle.
Improved ergonomics and optimized molar visibility.
KaVo launches the high-speed contra-angle MASTERmatic LUX M45 L with a unique head/knee angle combination for optimum visiblity in the molar area. The CoolHead technology and the optimized spray water of this new contra-angle handpiece ensure even greater patient safety during treatments.
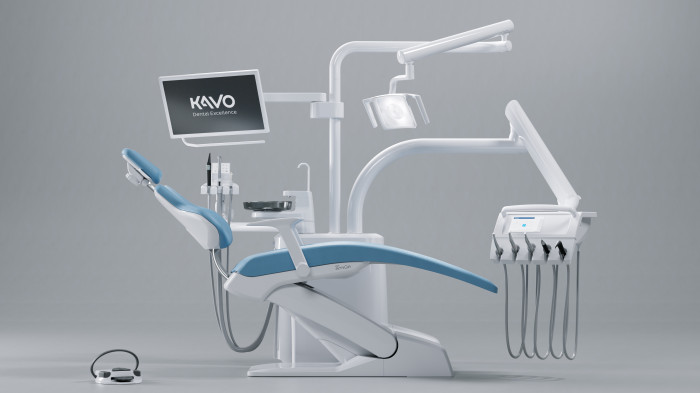
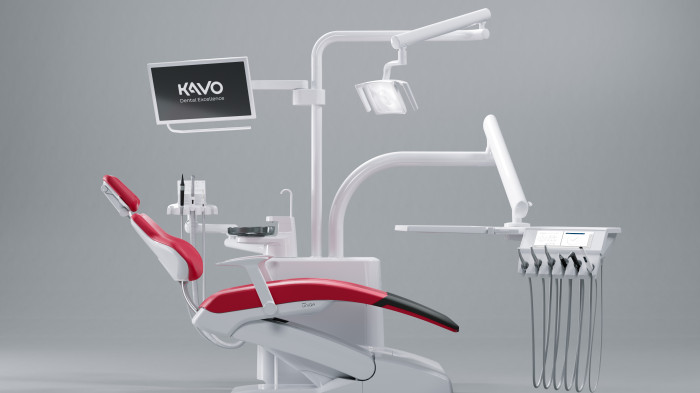
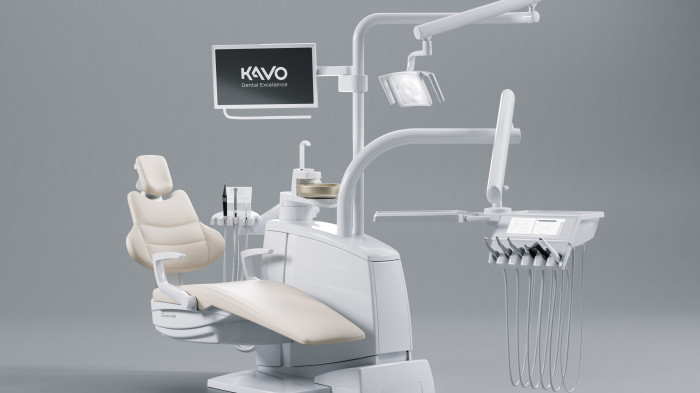
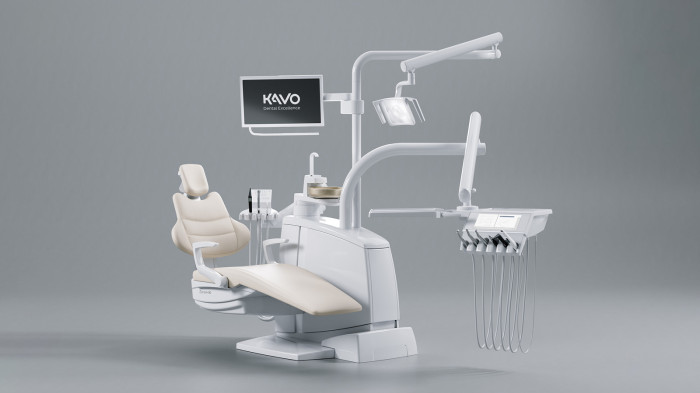
KaVo Configurator for Dental Chairs
A treatment unit as unique as you are – no problem with the KaVo treatment unit configurator!
Using the KaVo treatment unit configurator, you can assemble your new and unique treatment unit according to your specific needs and requirements.
Bookazines
A Bookazine is a mixture between product catalog and magazine. Our latest Bookazine contains product information and additonally topics like hygiene, ergonomics, practice interior, accessories and service.
Here you can download the KaVo Bookazines:
KaVo EXPERT Campaign
CoolHead technology – for even more patient safety.
The new KaVo CoolHead technology actively prevents excessive heating of the instrument head if the push button is pressed unintentionally during treatment while the burr is rotating.
KaVo exclusive high-tech ball bearings – for an even longer service life.
Feel free to contact us
Do you have any questions, would you like a quote, or would you like to discuss a specific issue directly?
Write to us using the contact form—we will get back to you as soon as possible.
KaVo has around 1,600 employees worldwide, all united by one common goal: to develop, manufacture and successfully market innovative, high-quality treatment units, instruments and product-related services.
We’re constantly implementing solutions to make modern dentistry better and delight our customers and partners all over the world. In short, we strive for nothing less than excellence.
repurchase rate
reflects high customer satisfaction.
locations
all over the world.
of all dental practices
in the western world use at least one KaVo product.
dedicated employees
worldwide.